In September 2016, our new Hepatitis B program (HOPE: Hepatitis B Overview and Program to trEat) launched. After months of negotiations, discussions and planning, Hepatitis B therapy was made available.
We are working alongside our partners at Hepatitis B Free (Australia) and Global Care Partners to support long-term access to anti-viral therapies for chronic Hepatitis B patients. The medicines used in the program (Tenofovir and Entecavir) suppress viral replication, prevent further inflammation and liver damage, and give hope for the preservation of both health and life while alleviating considerable suffering.
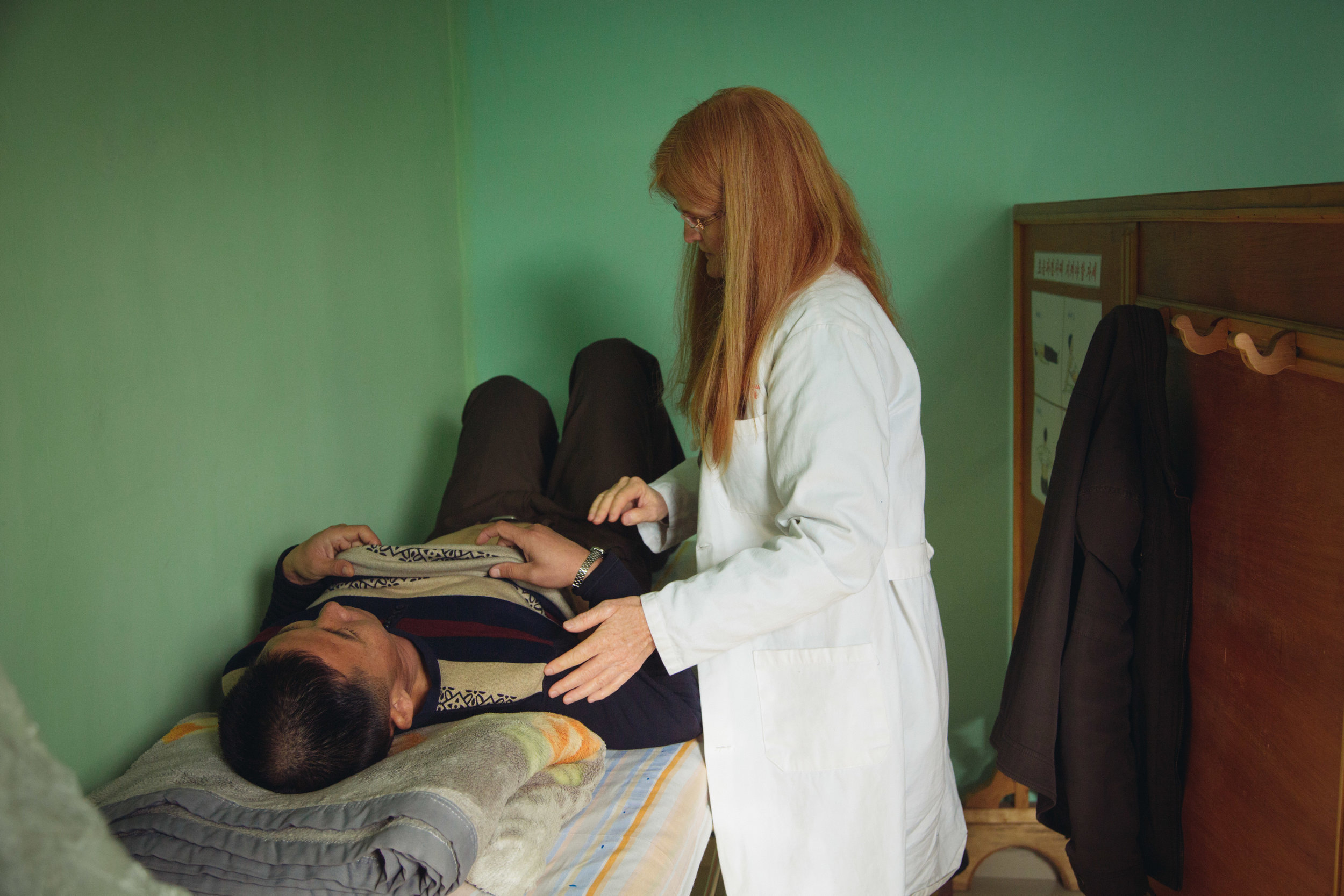
The HOPE program provides state-of-the-art medical care to Hepatitis B patients in a resource-challenged setting. Patients are evaluated through blood tests (immunology, chemistry, and hematology), ultrasound, fibroscan, physical exam, and health history. Each patient has his or her photograph taken for their ID card issued to them, signs a consent form, and is seen in clinic at least twice a year. So far, more than 600 patients have started treatment through the HOPE program, and we expect that more will be added throughout 2017.
HOPE Project Development Timeline
November
CFK was approached by MoPH and asked to begin hepatitis treatment facilities. From 2009-2015, we provide ongoing general support to five hepatitis care centers.
2009
March
Dr. Alice Lee (Co-founder of Hepatitis B Free [Australia]) makes her first visit with CFK to assess the hepatitis situation and explore possible next steps.
2014
May
After discussion and approval from MoPH, we initiate a project to test and vaccinate health care workers who are most at risk for contracting hepatitis B. The effort identifies a pressing need for antiviral therapy for people with chronic hepatitis B infection, launching an effort to develop a pilot program to address this need.
November
MoPH, CFK, and Hepatitis B Free (Australia) sign an MOU outlining the terms of a proposed long-term plan for antiviral hepatitis B therapy. Screening efforts and clinics are held in pilot locations at Kaesong and Pyongyang to begin identifying patients needing treatment. We held our first clinics and screened 384 patients for possible hepatitis B therapy which included running over 1,300 lab tests (which were completed at the NRL). Our joint team worked along with local hospital staff to examine 132 patients in Kaesong and 252 patients in Pyongyang.
2015
2016
march
Lab diagnostic tests are completed manually at the NRL clinical lab until the new labs are rebuilt in Kaesong and Pyongyang. Screening clinics continue at both facilities, and lab renovation work commences in Pyongyang.
May
Screening clinics continue in Kaesong and Pyongyang while lab renovation work continues in Pyongyang. A solar-powered water system is installed at Pyongyang #2 Hepatitis Hospital, and lab tests continue to be done manually at the NRL clinical lab.
In the summer of 2016, a one-year supply of antiviral medicines (Tenofovir and Entecavir) arrive in Pyongyang and are held until the HOPE team arrives.
September
While renovations of the lab at the Pyongyang #2 Hepatitis Hospital are completed and equipment is installed and renovation of the lab at the Kaesong #2 Hepatitis Hospital starts, clinics are held in both Kaesong and Pyongyang. The most urgent patients are started on antiviral therapy. In Pyongyang, 38 patients were started on treatment, and 35 patients were started in Kaesong.
november
Renovations of the Kaesong #2 Hepatitis Hospital are largely completed during this visit. Patient blood diagnostics are now processed in the newly renovated solar-powered lab at Pyongyang #2 Hepatitis Hospital. An additional 385 patients are started onto antiviral therapy in clinics held in Pyongyang and Kaesong during this visit.
2017
April
Kaesong #2 Hepatitis Hospital lab renovations were fully completed, and new equipment and supplies were installed. Meanwhile, a new automated chemistry analyzer was installed at Pyongyang #2 Hepatitis Hospital lab greatly increasing efficiency in blood chemistry diagnostics. Bar code scanning and automated processes were introduced into patient record keeping, improving efficiency and reducing the possibility for transcription errors. In-depth skills and knowledge transfer was delivered to medical, nursing, and lab staff at both facilities.
A new site was assessed for possible program expansion and clinics were held to give bi-annual checkups to patients who started on treatment in September 2016, as well as 44 additional patients who started in April.
June
Clinics were held in both Pyongyang and Kaesong. Bi-annual checkups for 353 patients started on treatment in November and 187 new patients were screened. Blood diagnostics were run on ( ) patient samples resulting in more than 6,600 re-portable tests. ( ) patients were started onto treatment in Kaesong and ( ) patients in Pyongyang. During this visit, CFK and MoPH agreed to expand the HOPE program into the South Hwanghae Province to include major improvements (well drilling, clean water system, lab renovations, etc.) at South Hwanghae #2 Hepatitis Hospital.
Significant training took place in June for the clinical and lab staff members at both hepatitis hospitals. Treatment decisions are made jointly in clinic by local and international physicians who volunteer in this project.
At the request of MoPH, we also visited a fourth provincial hepatitis hospital in a new region, where they hope to expand the hepatitis program in the future. Discussions are in process.
AUGUST / SEPTEMBER
Clinics were held in both Kaesong and Pyongyang from August 22 - 26. Our first patients were enrolled in the program and started treatment nearly one year ago, and they came back for their 12-month checkups. Many took time to say thank you – and there were many beautiful smiles, and “see you again soon.” So far in the program we have started 705 onto treatment, and screened over 1400 more.
In our screening for Hepatitis B, our protocol has required that we also test for Hepatitis C. Hepatitis C is also a virus that is transmitted through blood and is the other major cause of chronic liver disease throughout the world. Our program screens for hepatitis C as patients can be infected with both. So far we have found about 50 patients who are either positive for C or co-infected with both B and C; many are very sick with liver failure. We hope soon to be able to add hepatitis C treatment to our program.
NOVEMBER
Technical assistance continued at the Pyongyang and Kaesong Hepatitis Hospital labs, working side by side with local staff to process hundreds of blood samples and complete nearly 8,000 tests while improving local capacity for accurate diagnostics and treatment of hepatitis patients. New patient photos, diagnostic results and patient data were entered into our databases so that patient treatment records could be generated and printed in time for the clinical team’s arrival.
During the hepatitis B clinical team’s visits, our four medical doctors, working alongside our North Korean counterparts saw 172 patients in Kaesong over 2 days, and 420 patients in Pyongyang over 3 days. 134 patients were started on treatment (for the first time) and 6 or 12 month follow up exams were done on nearly 500 patients. Many patients are seeing significant improvement in their health and well-being as a result of antiviral therapy.
While patients waited to see the doctors they were able to read more about hepatitis and treatment through a patient information handbook developed by our team. Training of the staff also took place in both places.
“Be joyful in hope, be patient in affliction, be faithful in prayer.”